iHealth – Home COVID Tests (Carton of 180 Tests)
$1024.20/carton of 180 tests ($5.69/test)
CARTON PRICING
- 90 boxes per carton
- 2 tests per box
- 180 total tests
Please contact us for pallet pricing
QUICK SHIPPING! Ground, 2-day, and overnight (M-F) shipping available on in-stock test kits! All in-stock parcel shipment orders received before 1pm EST/10am PST ship same-day!
Contact Us to Purchase Fast & Secure Checkout
Description
- iHealth OTC COVID-19 Antigen Self-Test is intended for the qualitative detection of SARS-CoV-2 nucleocapsid protein antigens from individuals with or without symptoms or other epidemiological reasons to suspect a COVID-19 infection when tested twice over two or three days with at least 24 hours and not more than 48 hours between tests.
- EUA authorized for over-the-counter use
- 98.1% accuracy
- Native optimized mobile apps (app available in both Apple App Store and GooglePlay), intuitive UI
- Easier, automated reporting of results
- Reduced liability and costs
- Easy integration with existing employer portals/apps
- Detect SARS-CoV-2 nucleocapsid protein antigen
- PPA: 85-88%
- Instructions for Use at Home HERE
- Healthcare Provider Instructions for Use HERE
- Fact Sheet for Health Health Providers HERE
- Rapid results within 15 minutes
- Read about the difference between Antigen tests and Antibody tests
- Expiration Date: If you receive a box that has an expiration date that has passed, please do not be concerned. The state department has granted an extended expiration life FOR THIS PRODUCT. Please see extension letter HERE .
Pallet Info:
- 20 cartons/pallet
- 3,360 boxes/pallet
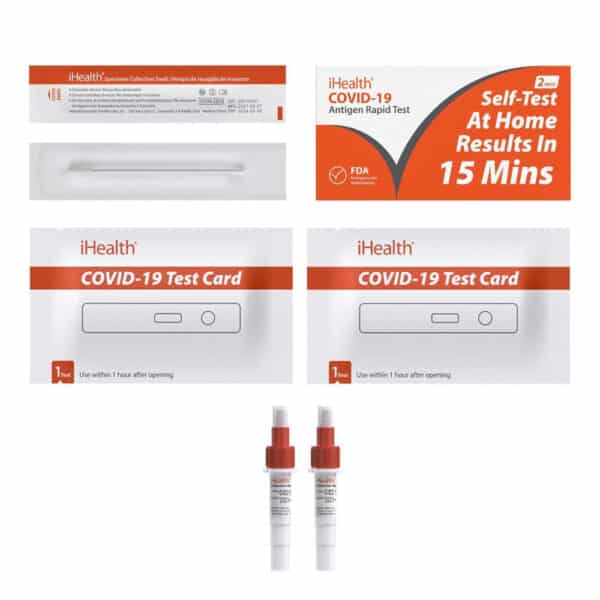
Contents Include:
- 2 Test devices: Foil pouched test device containing one test strip which is encased in plastic device cassette with a desiccant
- 2 Extraction vials and caps
- 2 Nasal swabs: swab for anterior nasal specimen collection
The iHealth antigen test kits contain two kits per box and are authorized for non-prescription home use. They are perfect for organizations and facilities that need to test staff or who need to make COVID-19 test kits available to check for potential exposures.
How Does the iHealth COVID-19 Antigen Rapid Test Work?
These tests can be used by anyone, not just medical personnel. In just a few minutes, these tests can tell someone whether they have COVID-19 antigens. Testing twice over three days with at least 24 hours between tests gives accurate results, and the test can be used by individuals with or without COVID-19 symptoms.
How to Use the iHealth Antigen Rapid Test
Each iHealth antigen test kit contains instructions. These instructions can also be downloaded from the iHealth COVID-19 Antigen Rapid Test app:
- Open the kit and remove all items from the test kit.
- Remove the swab from its package and place the entire tip of the swab, up to ¾ of an inch, into one nostril.
- Carefully move the swab in circles at least five times, brushing the insides of the nostril.
- Repeat with the other nostril using the same swab.
- Take the tube, tap it on a table and open the orange cap.
- Place the swab fully into the tube until it touches the bottom, and swirl it in the tube at least 15 times.
- Squeeze the sides of the tube to squeeze out more of the sample from the swab and then throw away the swab.
- Put the cap back on the tube and open the small white cap.
- Squeeze three drops from the tube into the sample well of the test card.
- Wait fifteen minutes to read the results.
Why Buy iHealth Antigen Rapid Tests?
This antigen test kit is a simple way to evaluate whether there have been exposures in the workplace and whether anyone needs to self-isolate. Regular testing is an important way for any organization today to keep its workplace safe.
Buy iHealth Antigen Rapid Tests in Bulk from SUNLINE Supply
SUNLINE Supply carefully vets every product we carry. With 24/7 shopping, low bulk prices and fast shipping on in-stock items, we’re your partner in COVID-19 at-home testing. Browse our products today.
Additional information
| Weight | 11 lbs |
|---|---|
| Dimensions | 12 × 12 × 13 in |
Expiration Dates:
- Expiration Date: If you receive a box that has an expiration date that has passed, please do not be concerned. The state department has granted an extended expiration life FOR THIS PRODUCT. Please see extension letter HERE.
EUA Info from the FDA:
- For use under an Emergency Use Authorization (EUA) only for the duration of the COVID-19 declaration justifying the emergency use of in vitro diagnostics (IVD), unless it is terminated or revoked by the FDA (after which the test may no longer be used)
- The iHealth™ COVID-19 Antigen Home Test is authorized for non-prescription home use with self-collected direct anterior nasal (nares) swab samples from individuals aged 14 years or older or adult collected direct anterior nasal swab samples from individuals aged two years or older.